Dr. Craig Brown

Position
Contact
Credentials
BA (Man), MSc (Calgary), PhD (Calgary)
Area of expertise
Synaptic plasticity, stroke, diabetes, in vivo imaging
Understanding the neural and vascular mechanisms of cortical plasticity and repair
Dr. Brown's neurobiology lab employs in vivo microscopic imaging technologies such as two-photon microscopy and functional imaging of neuronal and hemodynamic activity that allow the visualization of neuronal structures deep within the living brain or the processing of sensory information in real-time.
A central goal of Dr. Brown's research program is to use these experimental approaches to characterize the neurobiological mechanisms that allow the cerebral cortex to develop normally and change throughout life in response to new experiences (eg. learning, drug exposure) or pathology such as stroke or diabetes.
Donations support our students and faculty in their mission to produce internationally recognized advances in neuroscience knowledge.
Support Dr. Craig Brown's research.
OPPORTUNITIES:
The Brown lab is actively looking for talented new trainees at the graduate and post-doctoral level. Please contact Dr. Brown (brownc@uvic.ca) with your CV including reference list, unofficial transcripts and a research statement.
- Invasion of Phagocytic Galectin-3 expressing macrophages in the diabetic brain disrupts vascular repair
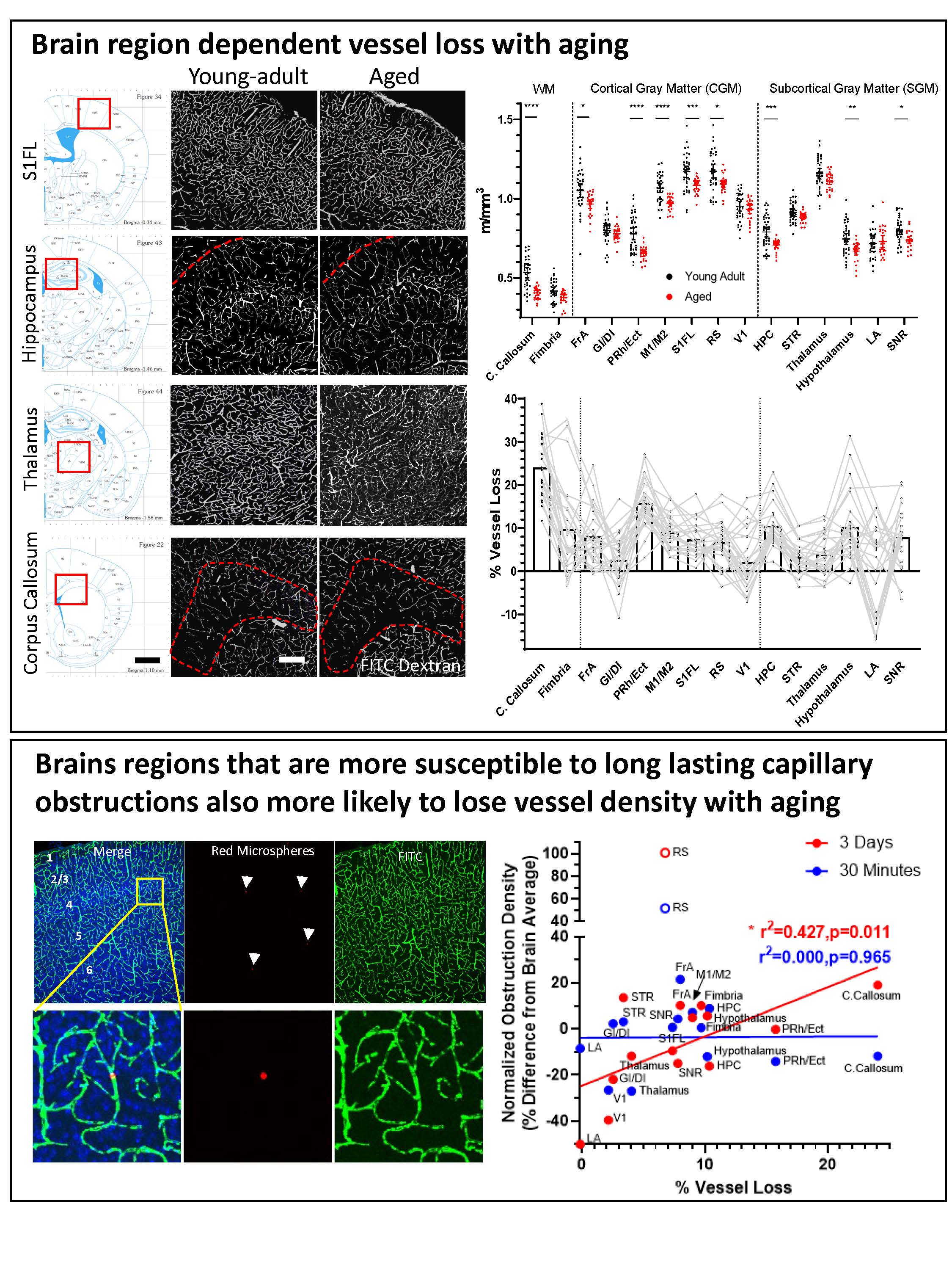
- Susceptibility to capillary plugging can predict brain region specific vessel loss with aging
- Suppressing IFN gamma stimulates microglial repair of microbleeds in diabetic brain
- Mapping and manipulating the fate of clogged brain capillaries
- “Optogenetic approach for re-wiring and restoring function in sensory circuits damaged by stroke”
- "Ultrastructure of blood-brain barrier breakdown after stroke"
- "Inhibition of VEGF signaling can improve stroke recovery in a co-morbidity dependent manner"
- "Diabetes augments in vivo microvascular blood flow dynamics after stroke"
- "Evaluating the impact of anesthesia on pre-clinical testing of neuroprotectants"
- "Diabetes impairs cortical map plasticity and functional recovery after stroke"
- "Nicotinic receptors modulate experience-dependent cortical plasticity"
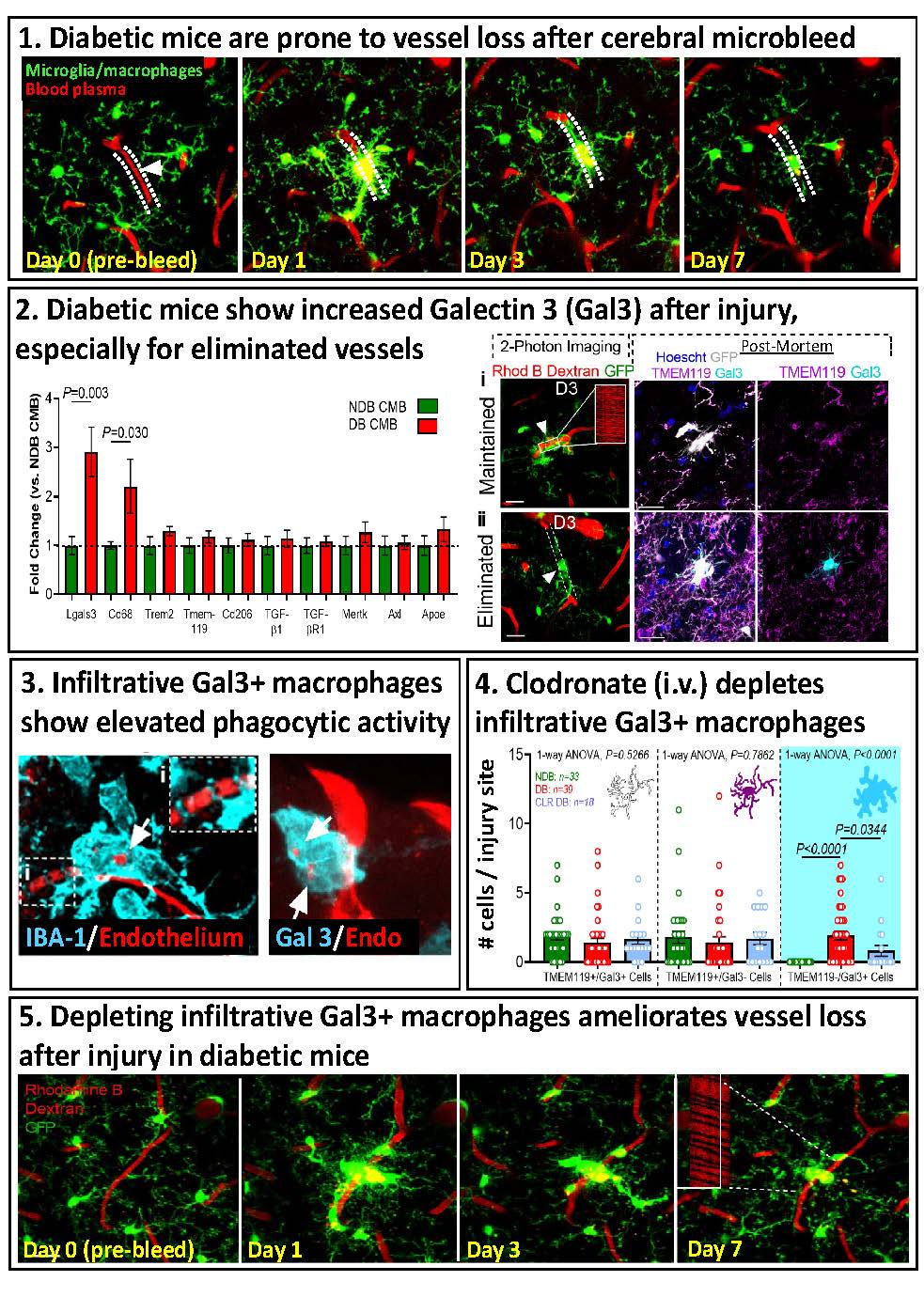
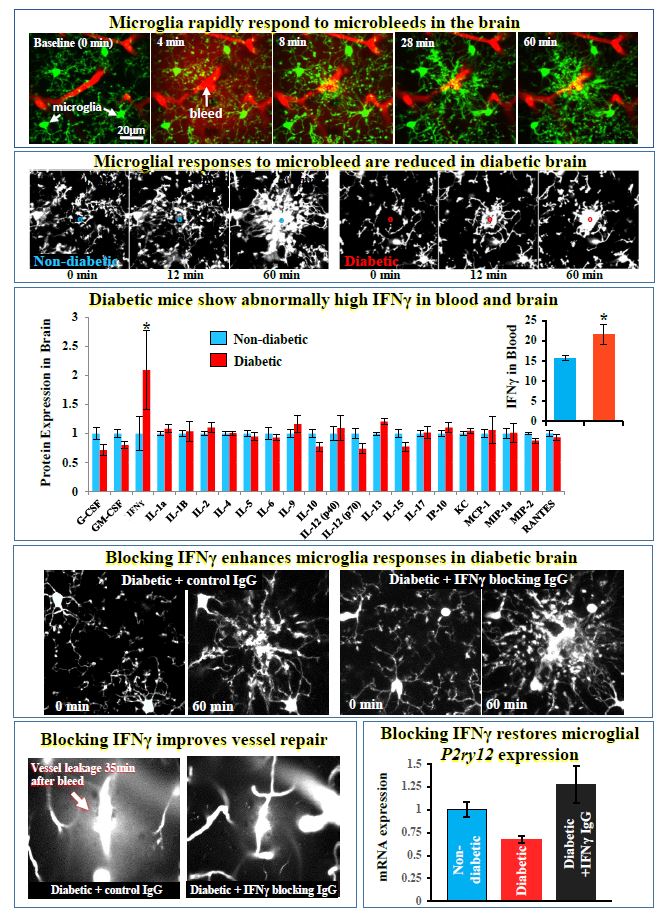
For more information: Taylor S, Mehina E, White E, Reeson P, Yongblah K, Doyle KP, Brown CE. 2018. Suppressing interferon gamma stimulates microglial responses and repair of microbleeds in the diabetic brain. Journal of Neuroscience Sep 12. pii: 0734-18. doi: 10.1523/JNEUROSCI.0734-18.2018. [Epub ahead of print]
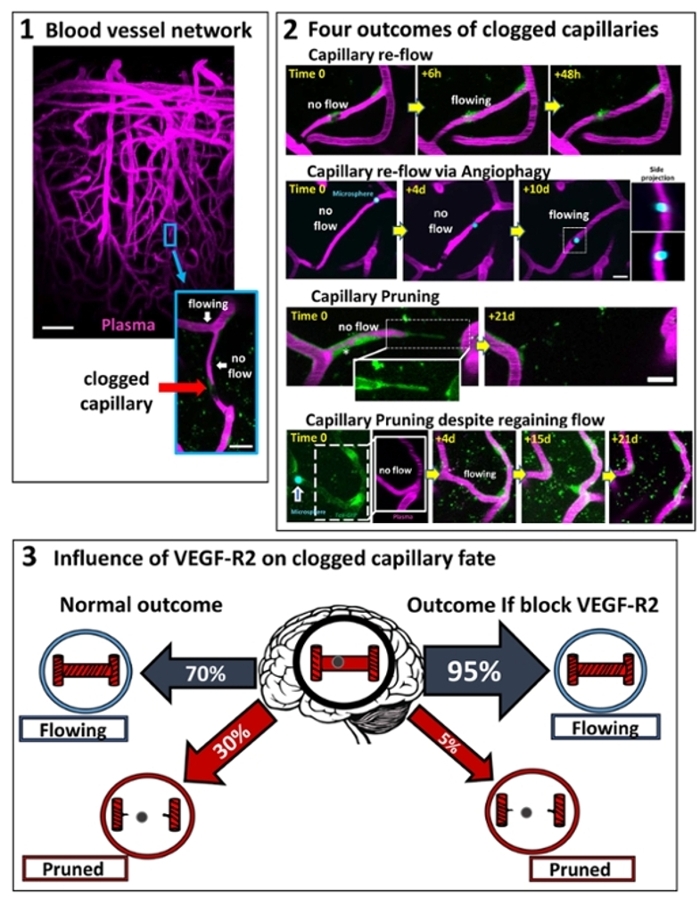
For more information see: Reeson, P, Choi K and Brown CE. 2018. VEGF signaling regulates the fate of obstructed capillaries in mouse cortex. eLIFE.
https://doi.org/10.7554/eLife.33670
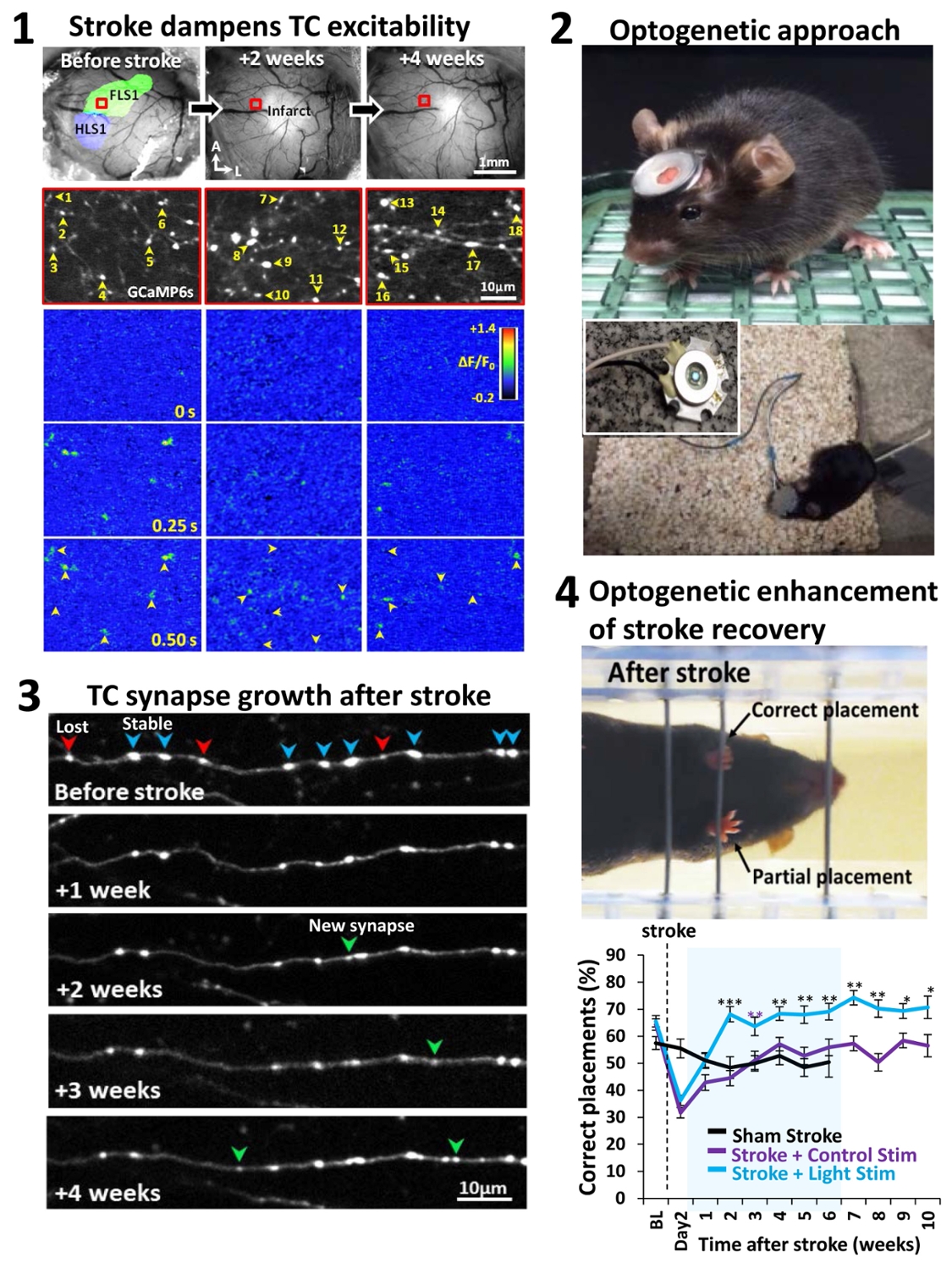
For more information see: Tennant KA, Taylor S, White E, Brown CE. 2017. Optogenetic rewiring of thalamocortical circuits to restore function in the stroke injured brain. Nature Communications Jun 23;8:15879. doi: 10.1038/ncomms15879.
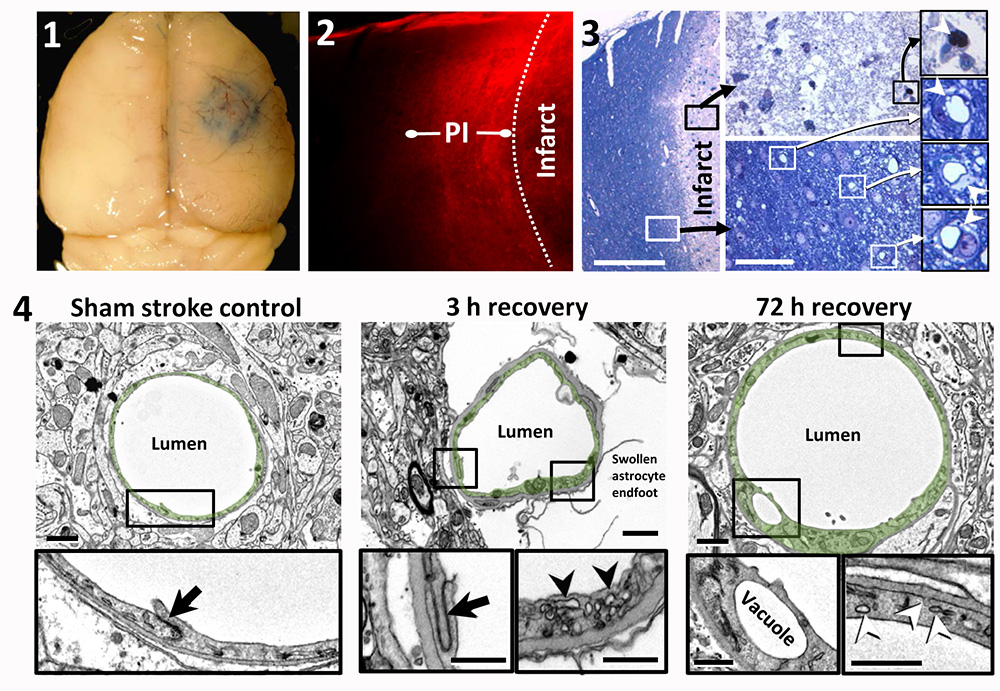
Focal ischemic stroke leads to a long-lasting increase in vascular permeability in the peri-infarct cortex of young adult and aged mice. 1. Whole-brain image showing Evans blue dye extravasation around the cerebral infarct 3 h after initiating photothrombosis. 2. Representative confocal image showing the halo of Evans blue fluorescence around the cerebral infarct. This halo of diffuse fluorescence usually extends 300–500 micrometers from the infarct border, which we refer to as ‘‘peri-infarct’’ (PI) cortex. Note that cortical areas more distant to the infarct exhibit little to no fluorescence. 3. A toluidine blue stained section showing the infarct core (above right) and peri-infarct region (below right) 72 h after stroke in an aged mouse. The vessels inside the infarct core were either plugged or unrecognizable. Open perfused microvessels were evident in the peri-infarct zone (below right). Close inspection reveals vacuolated endothelium and swollen astrocyte endfeet. 4. Electron microscopic images of capillaries in sham (left), and in the peri-infarct zone of 3 h (middle) and 72 h post-stroke cortical regions. High magnification views of the details of the endothelium reveal intact tight junctions (black arrows) and an increase in vesicles (arrowheads) within the endothelium. Vacuole formation in the endothelium was typical of capillaries in the peri-infarct zone.
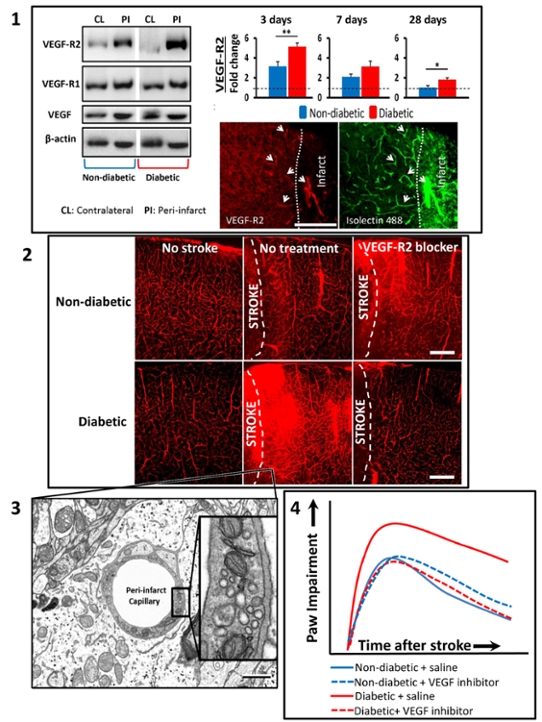
- Diabetes leads to abnormally high levels of VEGF-R2 expression in surviving areas next to the stroke (“peri-infarct regions”) . Fluorescent images show that VEGF-R2 expression is heightened in blood vessels next to the stroke
- Over-expression of VEGF-R2 in diabetic mice was associated with abnormally permeable blood vessels, which could be prevented with a VEGF-R2 inhibitor (semaxinib) or vessel specific genetic knockdown of VEGF-R2. Paradoxically, the same treatments in non-diabetic did not reduce vessel permeability.
- Electron microscopy revealed that vessel permeability was due to increased transport of blood constituents through the endothelial cells that line blood vessels (a trans-cellular route), rather than flowing between the cells through disrupted tight junctions.
- Inhibition of VEGF-R2 signalling improved the recovery of sensory-motor function after stroke, but only in diabetic mice. Journal of Neuroscience
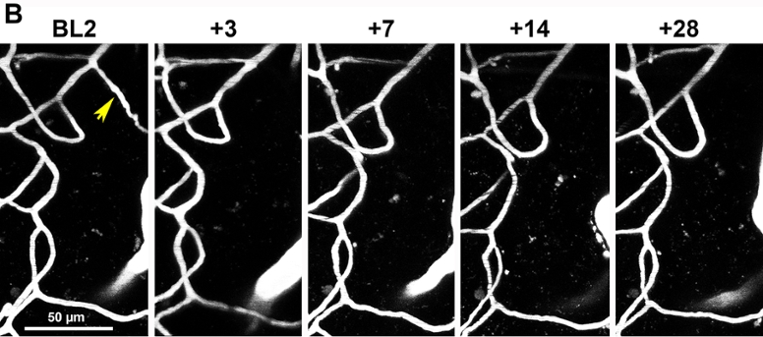
"longitudinal in vivo two-photon images of vascular networks in the peri-infarct cortex of diabetic mice subjected to ischemic stroke. For more details see: Tennant K and Brown CE. 2013. Diabetes augments in vivo microvascular blood flow dynamics after stroke. Journal of Neuroscience 33(49):19194-19204.
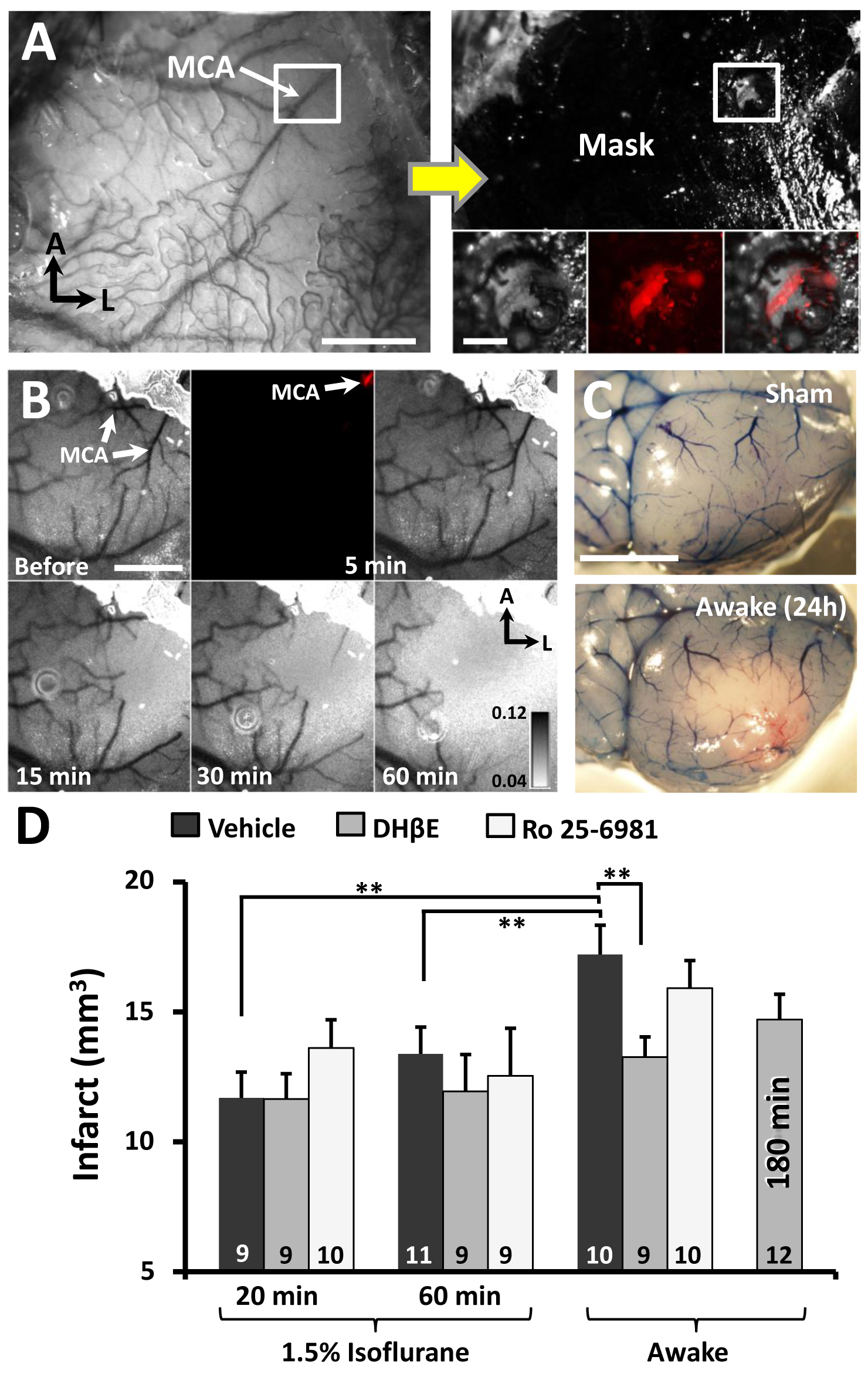
"(A) Photomicrograph of distal middle cerebral artery (MCA) through the intact mouse skull. Adjacent image shows skull afer masking procedure to restrict photoactivation to the distal MCA. Inset boxes show the segment of the MCA (labeled with Texas red dextran) that will be photoactivated. (B) Transcranial laser speckle contrast images of cortical blood flow at baseline and during 5, 15, 30 and 60 min of photoactivation. (C) surface vasculature labeled with Evans blue dye 24 hours after sham operation or stroke induction while awake. (D) Infarct volumes for awake or anesthetized mice receiving a glutamate receptor (GluN2B; Ro25-6981) antagonist or alpha-4 nicotinic receptors antagonist (DHβE).
Note the neuroprotective effect of the nicotinic antagonist is only evident in awake mice, perhaps because isoflurane is a known nicotinic receptor blocker. For more details, see Seto A, Taylor S, Trudeau D, Swan I, Leung J, Reeson P, Delaney KR, Brown CE.2014. Induction of ischemic stroke in awake freely moving mice reveals that isoflurane anesthesia can mask the benefits of a neuroprotection therapy.Frontiers in Neuroenergetics Apr 3;6:1.”
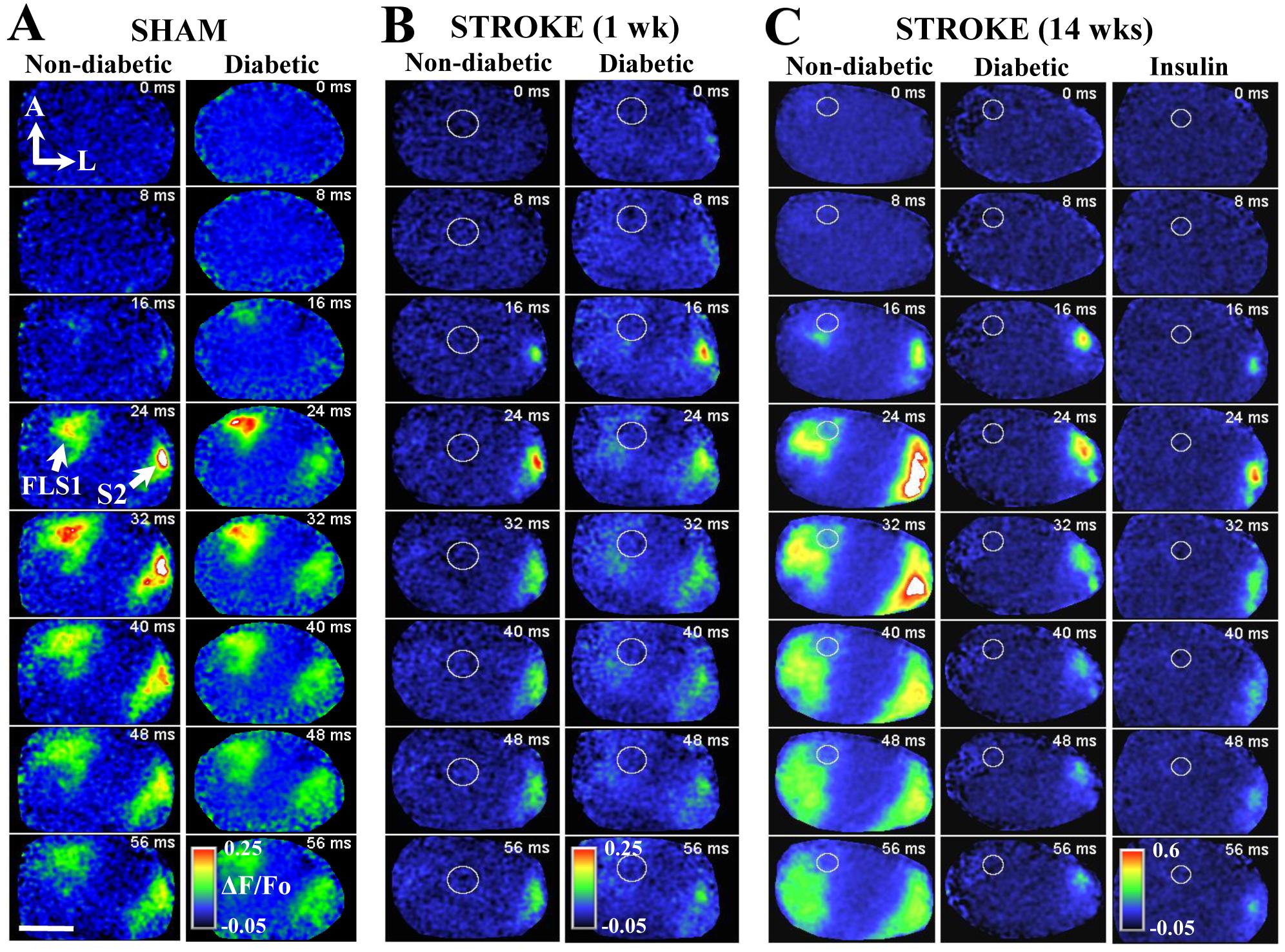
"In vivo voltage sensitive dye imaging reveals impaired cortical re-mapping of the forelimb sensory representation 14 weeks after stroke in diabetic and insulin treated mice. A: Montage shows forelimb evoked cortical depolarizations in sham stroke non-diabetic and diabetic mice. B: Montage shows the loss of forelimb-evoked cortical responses in regions next to stroke (indicated by white circle) in both non-diabetic and diabetic mice. After 14 weeks recovery from stroke, cortical responses to the forepaw re-emerge in peri-infarct cortex of non-diabetic. However, this re-mapping is significantly impaired in diabetic mice, as well as diabetic mice that had blood sugar levels controlled after stroke with insulin."
For more details see: Sweetnam D, Holmes A, Tennant K, Zamani A, Walle M, Jones P, Wong C, Brown CE. 2012 Diabetes impairs cortical plasticity and functional recovery following ischemic stroke. Journal of Neuroscience 32(15):5132-5143.
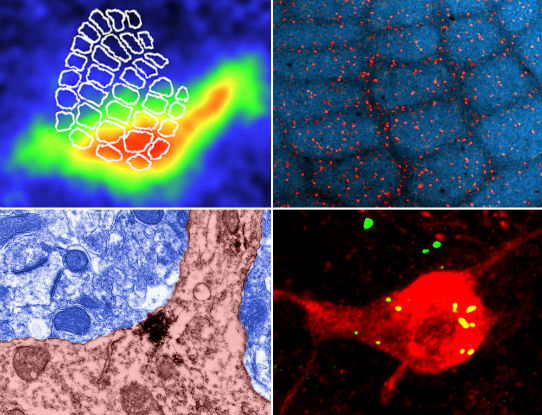
Top Left: In vivo voltage sensitive dye imaging of whisker-evoked depolarizations in the barrel cortex. Note that depolarizations (warm colors) are prominent in the barrel-field and secondary somatosensory cortex (right side of barrel-field). Top right: Low magnification confocal image showing the distribution of GABA-ergic neurons (red) in the barrel-field. Bottom right: High magnification confocal image reveals that YFP labelled alpha-4 nAChRs target GABA-ergic neurons in layer 4.Bottom left: Electron micrograph showing that alpha-4 nAChRs are expressed in neuronal cell bodies in putative peri/extra synaptic regions.
Brown CE, Sweetnam D, Beange M, Nahirney PC, Nashmi R. 2012. Alpha-4 nicotinic acetylcholine receptors modulate experience-based cortical depression in the adult somatosensory cortex. Journal of Neuroscience: 32(4): 1207-1219.
- Alejandra Raudales, Sorabh Sharma, Ben Schager, Dominique Hancock, Patrick Reeson, Manjinder Cheema, Jakob Korbelin, Craig E. Brown. 2024. Angiogenesis in the mature mouse cortex is governed in a regional and Notch1 dependent manner. Cell Reports.
- Sorabh Sharma, Manjinder Cheema, Patrick Reeson, Kelly A. Tennant, Roobina Boghozian, Ana Paula Cota, Tara P. Brosschot, Rachael D. Fitzpatrick, Jakob Körbelin, Lisa A. Reynolds, and Craig E. Brown. 2024. A pathogenic role for IL-10 signalling in capillary stalling and cognitive impairment in type 1 diabetes. Nature Metabolism Nov 4. doi: 10.1038/s42255-024-01159-9.
- Patrick Reeson and Craig E. Brown. 2024. Collaterals and stroke reperfusion: too few leads to too much. Neuron May 1;112(9):1378-1380. doi: 10.1016/j.neuron.2024.03.024.
- Boghozian R, Sharma S, Narayana K, Cheema M and Brown CE. Sex and interferon gamma signalling regulate microglia migration in adult mouse cortex in vivo. Proc Natl Acad Sci USA Jul 18;120(29):e2302892120. doi: 10.1073/pnas.2302892120.
- Motaharinia M, Gerrow G, Boghozian R, White E, Choi S, Delaney K, and Brown CE. 2021. Longitudinal functional imaging of VIP interneurons reveals sup-population specific effects of stroke that can be rescued with chemogenetic therapy. Nature Communications Oct 20;12(1):6112. doi: 10.1038/s41467-021-26405-6.
- Reeson P, Boghozian R, Cota AP, Brown CE. 2023. Optical opening of the blood brain barrier for targeted and ultra-sparse viral transfection of cells in mouse cortex. Cell Reports Methods Jun 2;3(6):100489. doi: 10.1016/j.crmeth.2023.100489.
- Reeson P, Schager B, Van Sprengel M, and Brown CE. 2022. Behavioural and neural activity dependent recanalization of clogged capillaries in the brain of adult and aged mice. Frontiers in Cellular Neuroscience May 26;16:876746. doi: 10.3389/fncel.2022.876746.
- Mehina EMF, Taylor S, Boghozian R, White E, Choi S, Cheema M, Korbelin J, Brown 2021. Invasion of Phagocytic Galectin-3 expressing macrophages in the diabetic brain disrupts vascular repair. Science Advances Aug 18;7(34):eabg2712. doi: 10.1126/sciadv.abg2712.
- Sharma S and Brown CE. 2021. Microvascular basis of Cognitive Impairment in Type 1 Diabetes. Pharmacology and Therapeutics. Jun 22:107929. doi: 10.1016/j.pharmthera.2021.107929.
- Reeson P and Brown CE. 2021. New Vistas in Stroke Research: Targeting the endothelium to restore blood flow. Circulation Research.
- Schager B and Brown CE. 2020. Susceptibility to capillary plugging can predict brain region specific vessel loss with aging. Journal of Cerebral Blood Flow and Metabolism Jan 5:271678X19895245. doi: 10.1177/0271678X19895245.
- Sanchez-Arias JC, Liu M, Choi CSW, Ebert SN, Brown CE, Swayne LA(2019) Pannexin 1 regulates network ensembles and dendritic spine development in cortical neurons. eNeuro 6: ENEURO.0503-18.2019 (original research).
- Taylor S, Mehina E, White E, Reeson P, Yongblah K, Doyle KP, Brown CE. 2018. Suppressing interferon gamma stimulates microglial responses and repair of microbleeds in the diabetic brain. Journal of Neuroscience Sep 12. pii: 0734-18. doi: 10.1523/JNEUROSCI.0734-18.2018. [Epub ahead of print]
- Reeson P, Choi K and Brown CE. 2018. VEGF signaling regulates the fate of obstructed capillaries in mouse cortex. eLIFE. https://doi.org/10.7554/eLife.33670
- Tennant KA, Taylor S, White E, Brown CE. 2017. Optogenetic rewiring of thalamocortical circuits to restore function in the stroke injured brain. Nature Communications Jun 23;8:15879. doi:10.1038/ncomms15879.
- Reeson P, Jeffery A, Brown CE. 2016. Illuminating the effects of stroke on the diabetic brain: Insights from imaging neural and vascular networks in experimental animal models. Diabetes 65:1779-1788.
- Nahirney, P.C., P. Reeson and C. E. Brown. 2016. Ultrastructural analysis of blood-brain barrier breakdown in the peri-infarct zone in young adult and aged mice. Journal of Cerebral Blood Flow and Metabolism 36(2): 413-25.
- Reeson P, Tennant KA, Gerrow K, Wang J, Weiser S, Thompson K, Lockhart K.L, Nahirney PC and Brown CE**. 2015. Delayed inhibition of VEGF signaling attenuates blood brain barrier breakdown and improves stroke recovery in a co-morbidity dependent manner. Journal of Neuroscience 35(13):5128-5143.
- Taylor S, Trudeau D, Arnold B, Wang J, Gerrow K, Summerfeldt K, Holmes A, Zamani A, Brocardo P and Brown CE**. 2015. VEGF can protect against blood barrier dysfunction, dendritic spine loss and spatial memory deficits in an experimental model of diabetes. Neurobiology of Disease 78:1-11.
- Seto A, Taylor S, Trudeau D, Swan I, Leung J, Reeson P, Delaney KR, Brown CE. 2014. Induction of ischemic stroke in awake freely moving mice reveals that isoflurane anesthesia can mask the benefits of a neuroprotection therapy. Frontiers in Neuroenergetics Apr 3;6:1.
- Tennant K and Brown CE. 2013. Diabetes augments in vivo microvascular blood flow dynamics after stroke. Journal of Neuroscience 33(49):19194-19204.
- Sweetnam D and Brown CE. 2013. Stroke induces long-lasting deficits in the temporal fidelity of sensory processing in the somatosensory cortex. Journal of Cerebral Blood Flow and Metabolism 33(1):91-96.
- Sweetnam D, Holme A, Tennant KA, Zamani A, Walle M, Jone P, Wong C, Brown CE.* 2012. Diabetes impairs cortical plasticity and functional recovery following ischemic stroke. Journal of Neuroscience 32(15):5132-5143. Full text PDF
- Brown CE, Sweetnam D, Beange M, Nahirnery PC, Nashmi R. 2012. Alpha 4 nicotinic acetylcholine receptors modulate experience-based cortical depression in the adult somatosensory cortex. Journal of Neuroscience 32(4):1207-1219. Full Text PDF
- Brown CE, Boyd JD and Murphy TH. 2010. Longitudinal in vivo imaging reveals balanced and branch specific dendritic remodeling of mature cortical pyramidal neurons after stroke. Journal of Cerebral Blood Flow and Metabolism 30(4):783-91.
- Brown CE, Aminoltejari K, Erb H, Winship IR and Murphy TH. 2009. In vivo voltage sensitive dye imaging in adult mice reveals that somatosensory maps lost to stroke are replaced over weeks by new structural and functional circuits with prolonged modes of activation within both the peri-infarct zone and distant sites. Journal of Neuroscience 29:1719-1734.
- Dahlhaus R, Hines RM, Eadie B, Kannangara T, Hines DJ, Brown CE, Christie BR, El-Husseini A. 2009. Over expression of the cell adhesion protein Neuroligin-1 induces learning deficits and impairs synaptic plasticity by altering the ratio of excitation to inhibition in the hippocampus. Hippocampus 20(2):305-22.
- Brown CE and Murphy TH. 2008. Livin' on the edge: Imaging dendritic spine turnover in the peri-infarct zone during ischemic stroke and recovery. The Neuroscientist 14:139-146.
- Brown CE, Wong C and Murphy TH. 2008. Rapid morphological plasticity of peri-infarct dendritic spines after focal ischemic stroke. Stroke 39:1286-1291.
- Liu RR, Brown CE, Murphy TH. 2007. Differential regulation of cell proliferation in neurogenic zones in mice lacking cystine transport by xCT. Biochemical Biophysical Research Communications 364:528-533.
- Brown CE, Li P, Boyd JD, Delaney KR and Murphy TH. 2007. Extensive turnover of dendritic spines and vascular remodelling in cortical tissues recovering from stroke. Journal of Neuroscience 27:4101-4109.
- Flynn C, Brown CE, Galasso SL, McIntyre DC, Teskey GC, Dyck RH. 2007. Zincergic innervation of the forebrain distinguishes epilepsy- prone from epilepsy-resistant rat strains. Neuroscience 144:1409-1414.
- Brown CE and Dyck RH. 2005. Modulation of synaptic zinc in barrel cortex by whisker stimulation. Neuroscience 134:355-359.
- Brown CE and Dyck RH. 2005. Retrograde tracing of the subset of afferent connections in mouse barrel cortex provided by zincergic neurons. Journal of Comparative Neurology 486:48-60.
- Brown CE and Dyck RH. 2004. Distribution of zincergic neurons in the mouse forebrain. Journal of Comparative Neurology 479:156-167.
- Schuurmans C, Armant O, Nieto M, Stenman JM, Britz O, Klenin N, Seibt J, Brown C, Tang H, Cunningham JM, Dyck R, Walsh C, Campbell K, Polleux F, Guillemot F. 2004. Sequential phases of neocortical fate specification involve Neurogenin-dependent and independent pathways. EMBO Journal 23:2892-2902.
- Brown CE and Dyck RH. 2003. An improved method for visualizing the cell bodies of zincergic neurons. Journal of Neuroscience Methods 129:41-47.
- Brown CE and Dyck RH. 2003. Experience-dependent regulation of synaptic zinc is impaired in the cortex of aged mice. Neuroscience 119:795-801.
- Brown CE, Seif I, De Maeyer E, Dyck RH. 2003. Altered zincergic innervation of the developing primary somatosensory cortex in monoamine-oxidase A knockout mice. Developmental Brain Research 142:19-29.
- Brown CE and Dyck RH. 2002. Rapid, experience-dependent changes in levels of synaptic zinc in primary somatosensory cortex of the adult mouse. Journal of Neuroscience 22:2617-2625.

 |
Roobina Boghozian, Post-doc |
 |
Sorabh Sharma, Post-doc My primary interest is to study how diabetes alters the neurological function. Recently, increased prevalence of neurodegenerative diseases have been reported in patients suffering from Type 2 diabetes. However, the underlying mechanisms of association between these conditions are still elusive. During my doctorate, I explored the possible involvement of epigenetic modifications in diabetes induced neurodegeneration. We reported that dysregulation of epigenetic pathways leads to cognitive deficit and amyloid-β accumulation in hippocampus of mice. We also examined the therapeutic potential of histone deacetylases inhibitors, during these co-morbid conditions. As a Post doc in the Brown lab, I will be studying how diabetes affects the rates of capillary obstruction and/or pruning. Further, we will focus to elucidate mechanistic understanding of how obstructions are cleared. This could be used in a therapeutic manner to preserve brain micro-circulation and function throughout the lifespan. |
 |
Eslam Mehina, Ph.D., Neuroscience Graduate Program Research Interests: My project aims to decipher the role of microglia in responding to damaged microvessels in the brain, and in contributing to their repair and ultimate fate after hemorrhagic insults. Diabetic patients are at increased risk of suffering from cerebrovascular disease, so I am studying whether microglial dynamics and responses are disturbed in mice with a model of insulin-dependent diabetes. If I find that they are, I will also be investigating means by which these changes can be reversed to prospectively reduce the risk of developing these pathologies. |
 |
Mohammad Motaharinia, M.Sc., Neuroscience Graduate Program |
 |
Ben Schager, M.Sc., Neuroscience Graduate Program |
| Julia Templeman, Volunteer/ Honours Student |
This could be you! We are currently seeking applications for new lab members. Please contact Dr. Brown for details at brownc@uvic.ca.
- Heart & Stroke Foundation of BC & Yukon - Peripheral nerve stimulation to enhance recovery from ischemic cerebral stroke.
- Canadian Institute of Health Research (CIHR) - CIHR New Investigator Award - Imaging the impact of diabetes on brain function and recovery from stroke.
- NSERC Discovery Grant - Imaging rapid, use-dependent plasticity in adult somatosensory cortex.
- NSERC Equipment Grant - Equipment for imaging functional domains in the cerebral cortex.
- Canadian Foundation for Innovation (CFI) - Two-photon microscope for imging neuronal and vascular circuit assembly and remodeling in the living brain.
- Michael Smith Foundation for Health Research - Career Investigator Award - Research focus on understanding the mechanisms of experience and injury based cortical plasticity.
- Canadian Institute of Health Research (CIHR) Operating Grant - 5 year operating grant 2013-2018 - Impact of diabetes on brain function and recovery from ischemic stroke.
Kelly Tennant, PhD - Post Doctoral Fellow
September 2011 - January 2018
 |
Patrick Reeson, Ph.D., Neuroscience Graduate Program Research Interests: Diabetics are more likely to suffer a stroke, and when they do their prognosis for recovery is significantly worse. Previous work in the lab has established that the surviving cortical tissue in the diabetic brain is impaired in its ability to adapt to facilitate recovery. Given the importance of the cerebraovasculature to neuronal function and the widespread vascular pathologies associeated with diabetes, Patrick's work aims to understand how vascular dysfunction in the diabetic brain after stroke impairs plasticity and recovery. Patrick utilizes confocal and 2 photon microscopy to image the cerebrovasculature as well as molecular methods to study key angiogenic and vascular permeability factors such as VEGF. |
 |
Kim Gerrow, Ph.D. Post Doctoral Fellow Research Interests: During stroke recovery, pyramidal neurons in the peri-infarct cortex undergo structural re-arrangements, and in contrast, very little is known about how stroke affects the morphology of cortical interneurons. Many interneurons release GABA and/or peptides that regulate cortical excitability, which is profoundly disrupted after stroke. Of note, interneurons expressing vasoactive intestinal peptide (VIP) specialize in inhibiting other classes of inhibitory cortical neurons, such as those expressing parvalbumin (PV) and somatostatin (SOM). Using longitudinal in vivo imaging and Cre-dependent transgenic mouse lines, we are currently investigating how stroke alters the growth and stability of dendritic arbors, spines and axons in interneurons. |
| Stephanie Taylor - MSc Student January 12, 2017, Stephanie successfully defended her thesis titled: Diabetes impairs the microglial response to cerebral microbleeds |
|
Angela Seto - MSc Student June 10, 2013, Angela successfully defended her MSc thesis titled: Role of the a4B2 nicotinic acetylcholine receptor in stroke recovery. |
|
Andrew Sweetnam-Holmes - MSc Student August 15, 2013, Andrew successfully defended his MSc thesis titled: Diabetes exacerbates the loss of dendritic spines after ischemic stroke. |
|
Dani Sweetnam-Holmes - MSc Student November 2011, Dani successfully defended her MSc thesis titled "Diabetes impairs cortical plasticity and functional recovery following ischemic stroke". Dani is continuing her education in medicine. |
